STEMart, a US-based provider of comprehensive services for all phases of medical device development, has announced the launch of its Rapid Endotoxin Testing Services to help medical device manufacturers quickly and accurately assess the endotoxin levels in their products.
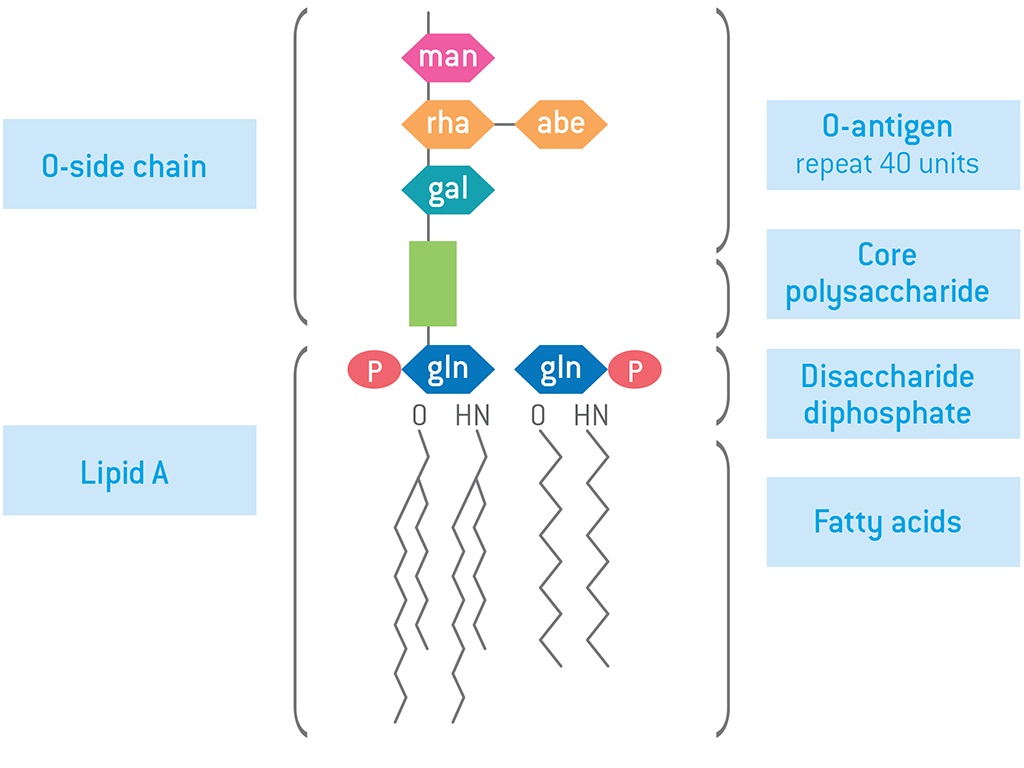
Bacterial endotoxins are lipopolysaccharides (LPS) bound to the bacterial cell wall and released when the bacteria rupture or break down. They consist of three major components, O antigen, core oligosaccharide, and lipid A molecule. Endotoxin contamination of blood can cause a variety of adverse biological effects, including inflammatory responses, immune system invasion, septic shock, and adult respiratory distress syndrome.
According to the FDA guidance document ‘Pyrogen and Endotoxins Testing: Questions and Answers’, the endotoxin limit for a medical device depends on the intended use of the device and who the device is intended to come into contact with. For products in contact with the cardiovascular and lymphatic systems, the limits are 0.5 EU/mL or 20 EU/device. For devices in contact with cerebrospinal fluid, the limit is 0.06 EU/mL or 2.15 EU/device. Low endotoxin limits are recommended for devices in contact with the intraocular environment.
Measuring endotoxin in various medical device materials is critical to assessing product safety and reducing risk. With increased regulatory scrutiny of medical device endotoxin in national and international approval processes, endotoxin testing has become increasingly important for medical device products, especially injectable and implantable products. Currently, the primary method used to detect and quantify endotoxin or LPS is the Limulus Amebocyte Lysate (LAL) assay. In addition, photometric and mass spectrometry (MS)-based techniques are applied for some assays.
STEMart now offers Rapid Endotoxin Testing services for the rapid quantitative analysis of endotoxins to help manufacturers make fast, reliable decisions about the safety of medical devices. The testing methods include a range of endotoxin testing instruments, FDA-licensed reagents and software, and a proprietary cartridge. With these rapid testing methods, customers can improve sample management, reduce testing time, and accelerate product manufacturing.
STEMart offers comprehensive sterility testing for sterile, non-pyrogenic products. STEMart’s extensive expertise in microbiology and sterility testing provides a full-service experience that helps manufacturers meet regulatory goals and minimize compliance risk. Whether developing a new device or improving an existing one, STEMart has the experience to guide clients through the design, engineering, manufacturing and regulatory challenges unique to the medical device development process.
If you have additional questions about Rapid Endotoxin Testing for medical devices or would like more information about other medical device services, please visit https://www.ste-mart.com/rapid-endotoxin-testing-for-medical-devices.htm.
About STEMart
STEMart is an industry-leading eCommerce platform incorporated with an extensive global footprint and a broad portfolio of more than 10,000 products. It aims to provide better lab materials, medical instruments and consumables, excellent technologies, and high-quality services to global customers in the fields of science, technology, and engineering, from the discovery stage upward to the manufacturing process. STEMart is dedicated to enhancing research and biotech production with simpler and safer protocols to access better health worldwide.